
For example SF4 is apparently an AB4 type molecule but. The bond angle indicated is exactly 109. 
For example, carbon dioxide and nitric oxide have a linear molecular shape. Which of the following is the best statement about the bond angle indicated by the double arrow in the diagram. Types of molecular structure A bond angle is the geometric angle between two. When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry (actual. The total number of electron pairs, both bonding pairs and lone pairs, leads to what is called the electron domain geometry. The molecular geometry is the shape of the molecule. Transcribed Image Text: Part A Below is a cartoon drawing of a seesaw molecular geometry which is based upon the trigonal bipyramidal electron group geometry. The molecular geometries of molecules change when the central atom has one or more lone pairs of electrons. The electron-pair geometry provides a guide to the bond angles of between a terminal-central-terminal atom in a compound. Molecular geometry is the name of the geometry used to describe the shape of a molecule. Seesaw molecular geometry Disphenoidal or seesaw (also known as sawhorse 1) is a type of molecular geometry where there are four bonds to a central atom. Well use the example of SF4 to understand the molecular shape. of more atoms, the shape depends on the number and type of electrons (bonding. When all of these regions are bonding, the. In this video we’ll look at the Seesaw Molecular Geometry and Bond Angles. three-dimensional shape of a molecule or polyatomic ion. Electronegative ligands such as F will always go to the axial sites.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. See-saw geometry is just like trigonal bi-pyramidal geometry that results due to dsp3 hybridisation. Molecular geometry is the name of the geometry used to describe the shape of a molecule. Transcribed Image Text: Part A Below is a cartoon drawing of a seesaw molecular geometry which is based upon the trigonal bipyramidal electron group geometry. The seesaw shape has 5 electron density regions (trigonal bipyramidal), with four bonding pairs and one lone pair. 
Both are given by the VSEPR theory, but they are not the same thing. 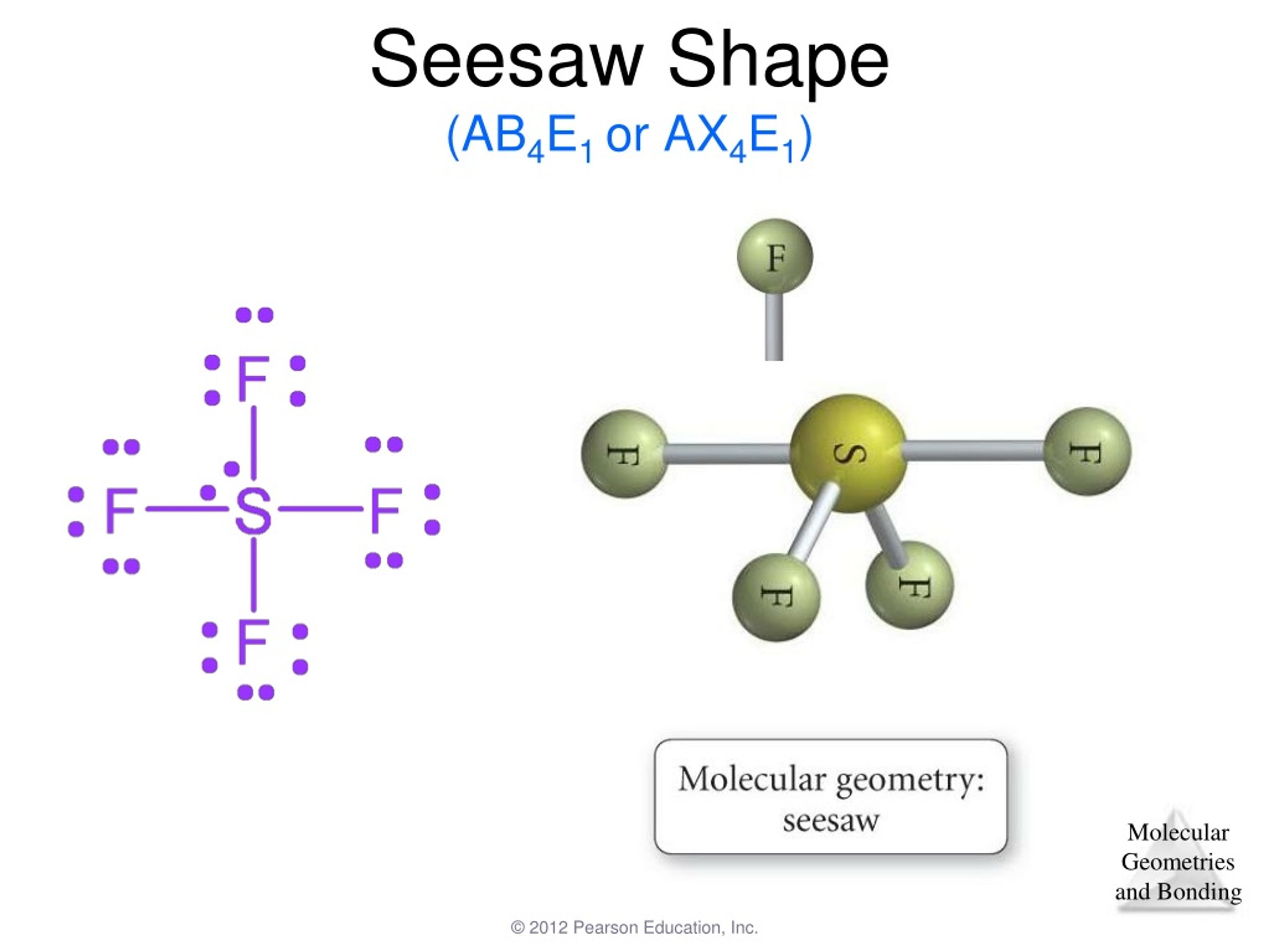
In general, by this reasoning, lone pairs and electropositive ligands such as CH 3 will always prefer the equatorial sites in the trigonal bipyramidal geometry. shape ENDORSED Geometry refers to the arrangement of the electron groups, while shape refers to the overall shape of the molecule. The activity involved science students preparing origami models of triangular bipyramidal and seesaw molecular geometries. = 0.867 \:bond (formal \: charge = -0.122)\)īecause fluorine is more electronegative than a lone pair, it prefers the axial site where it will have more negative formal charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
